Peeling Back Complexity: How We’re Simplifying African Healthcare Regulation, One Layer at a Time
The barriers to access weren’t always about innovation or cost — they were often about regulatory complexity.

“Most people stop at the first solution. But if you keep going, you can arrive at something very elegant and simple.” — Steve Jobs
When I first came across that quote, I felt it described not just product design, but my entire entrepreneurial journey with Infinity Health Africa.
In 2023, when I began shaping the vision for Infinity Health Africa, our mission was simple in theory — but incredibly complex in practice: to improve access to innovative healthcare products and services across Africa. For years, I had seen how the lack of access drove up costs, pushed people toward medical tourism, and stalled innovation. We wanted to bridge that gap by supporting innovators with data, regulatory insight, and launch support.
But the first version of that dream was too big, too abstract — and far too difficult to explain to investors.
We didn’t originally plan to focus on regulatory. What I wanted most to fix was the lack of reliable, on-the-ground data. Having spent years in product management and launch roles for Big Pharma and digital health companies across Sub-Saharan Africa, I knew firsthand how data gaps lead to poor execution. On paper, a product would look viable — but in-market, the story was entirely different. The data rarely matched the reality.
So instead of guessing, we decided to listen.
I personally spoke with every single customer lead we got in our first year — and still do when I can. Those conversations were eye-opening. Almost every innovator we met wanted to enter African markets — but was stuck at the regulatory level.
One moment crystallized everything for me. In 2020, my father needed a pacemaker — urgently. It wasn’t available in Nigeria, but I was living in Kenya then and could source one there. Years later, during a customer conversation, I asked a manufacturer why such a device wasn’t registered in Nigeria. The answer? They couldn’t navigate the regulatory environment.
That’s when it hit me: The barriers to access weren’t always about innovation or cost — they were often about regulatory complexity.
Learning by Serving
We didn’t learn all this in a lab or strategy session — we learnt by serving customers.
Sometimes painfully. Sometimes manually. But always with the goal of deeply understanding the real pain points, not the ones we imagined.
We processed documents by hand. We sat in queues with clients. We joined their regulatory battles — from misfiled applications to opaque feedback. And by doing so, we learnt where the process breaks down. We learnt where regulators miss it, where businesses give up, and where systems fail.
For software to work for people, it must begin with people. We walked the paths our users walked and found the lessons embedded in them.
This was never about tech for tech’s sake. It was about building the most efficient and simple software possible, grounded in the real-world workflows of the people it serves.
Building ONBOARD: Simplicity After Complexity
So we pivoted. We decided to build a system that could simplify regulatory compliance for healthcare companies across Africa.
It hasn’t been easy. African regulation is notoriously fragmented — different rules, different expectations, and very little clarity. Many regulators lack digital infrastructure. Many businesses don’t know who to ask or where to begin. The very people trying to bring innovation into our markets often give up before they start.
Instead of tackling everything at once, we started with the questions our customers asked the most. The most common pain point?
“I don’t know what the requirements are — or how to prepare the right documentation.”
Many companies simply didn’t understand the basic steps of regulatory compliance. Even when guidelines were available, they were buried in dense SOPs or outdated documents. And more often than not, businesses were left to figure it out on their own — leading to delays, rejections, or giving up entirely. But it wasn’t just about knowing what to submit — it was about knowing how to prepare quality documentation that met the standards of African regulators.
So we built a solution.
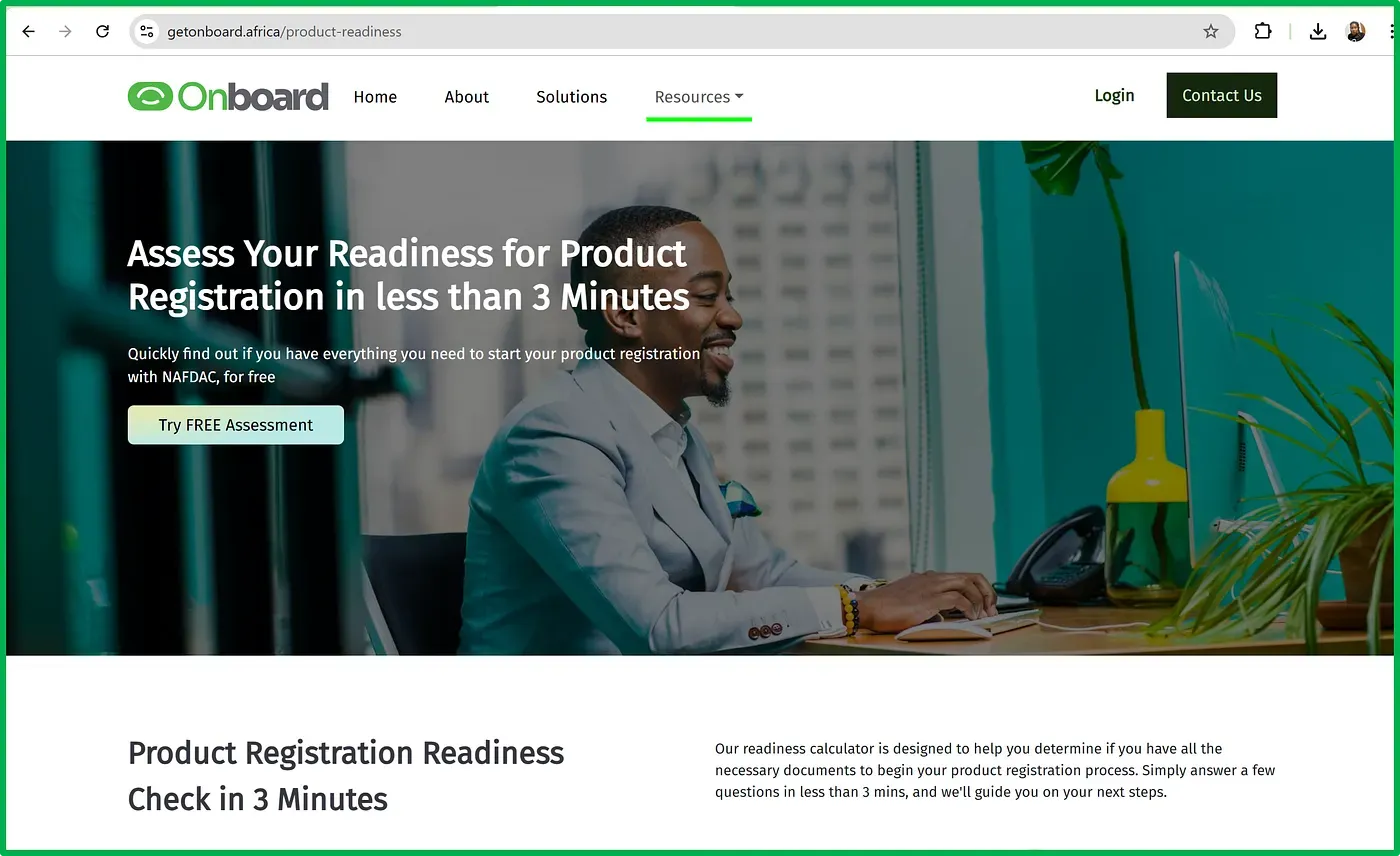
We took the dense and often unreadable SOPs from regulators like NAFDAC and gamified them. The result? A Product Readiness Calculator — a simple, guided tool that not only shows you what the requirements are, but also teaches you what good documentation looks like, where to find the necessary information, and how to get support if you’re stuck.
It’s like a regulatory GPS — guiding companies from confusion to clarity.
Since its rollout, more than 150 businesses have used the tool. And the feedback has been consistent:
“This saved us months of confusion.”
But we didn’t stop there. We engaged directly with regulators. We still do. Co-creating tools that not only support industry players but also make life easier for regulators themselves. After all, true innovation can’t happen without both sides at the table.
It’s Still Day One
ONBOARD is still evolving — and so is Infinity Health Africa. We’re proud of what we’ve built so far, but we know there’s still a long road ahead. We’re learning every day, iterating fast, and staying close to the people we serve.
The African healthcare market doesn’t need more complicated systems. It needs clarity. It needs tools that work. It needs partners who listen.
What started out as a messy, ambitious vision is slowly becoming something elegant. That’s what happens when you keep peeling back the layers.
And we’re just getting started.
Research-driven writing on healthcare, technology, policy, and the systems that shape care across Africa.












Member discussion