The "PR Gap" in Medicine: Why Your Social Media "Breakthrough" Isn't a Cure (Yet)
We are living in an era of viral science. But as funding dries up and AI speeds up, the line between "proof of concept" and "proven cure" is becoming dangerously blurred.
A video of a white powder stopping a deep bleed in one second goes viral on X. A headline claims a "cancer-killing pill" just cleared its first trial. A post announces that AI has found a cure for Alzheimer's.
If you feel like we're living in a golden age of medical miracles, you're not alone. But if you feel like these "miracles" never actually make it to your local pharmacy, you're also not wrong.
In 2026, we are witnessing a fascinating, albeit painful, shift in the geopolitical and economic landscape of healthcare. Scientists are "locking in" like never before, but they are also being forced into a new role: the marketer.
The Funding Hunger Games
The reality of 2026 is that grant funding is diminishing globally. Scientific institutions and academics can no longer rely solely on the slow, quiet trickle of government checks. To keep the lights on and the lab running, they now have to engage in aggressive PR.
This creates what I call the "PR Gap." When a researcher or news media announces a revolutionary treatment too early in its development cycle, the goal isn't just to save lives. It's also to capture the attention of funders.
The problem? The public sees these announcements as finished products. They aren't.
The reason you are hearing so many breakthroughs in medicine right now is because scientists and academics have to engage in PR to raise funding to continue their work. The public considers these announcements real and builds hope around them. But it is not real yet. Lots of these trials fail. It is not uhuru yet.
I wish scientists didn't have to do this to get funding. But this is the world we are in.
The Long Walk: Phase 1 vs. Phase 3
Most "breakthroughs" you see on your feed are only at Phase 1 or Phase 2. This is the scientific equivalent of a "vibe check."
To actually secure approval for a drug or medical product, it must survive the grueling "Valley of Death" that is a Phase 3 trial.
The Drug Approval Pipeline: What Each Stage Actually Means

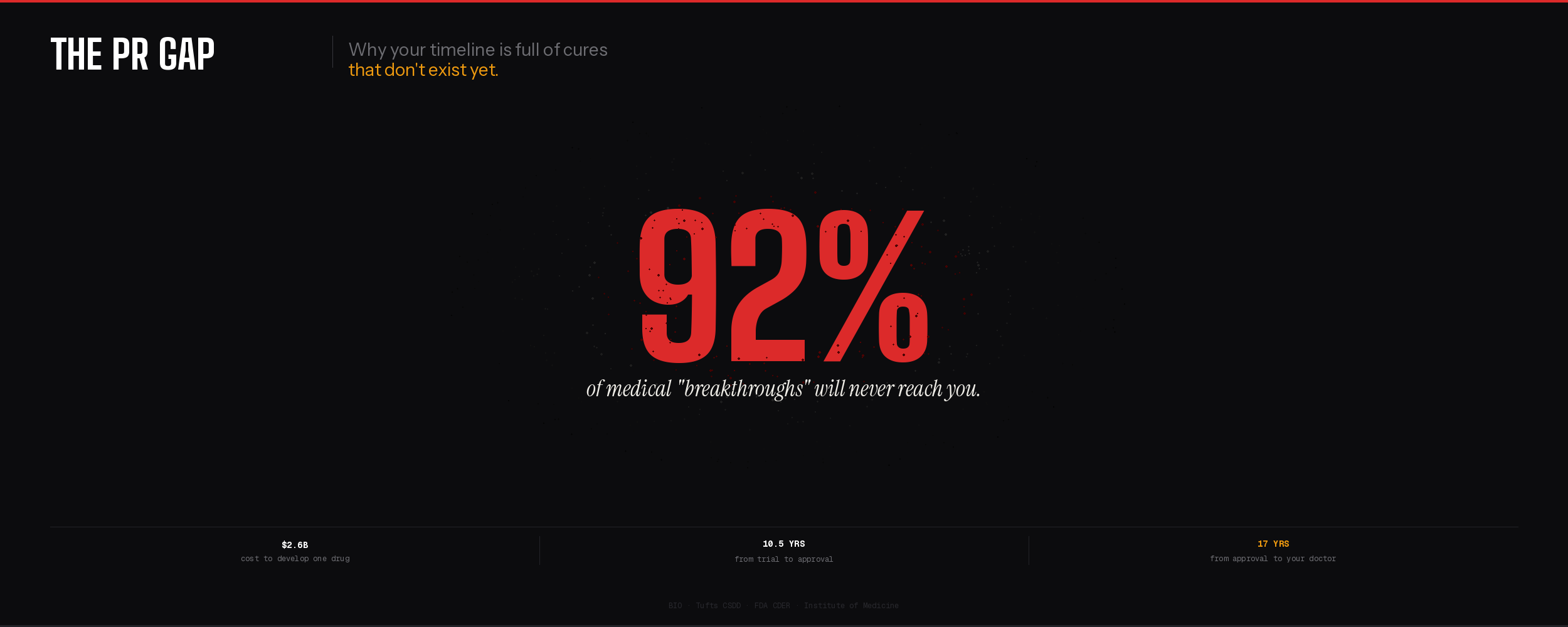
Here is the sobering math: according to BIO, the average likelihood of approval for all drugs entering Phase 1 is only 7.9%. That means more than 9 out of every 10 drugs you see celebrated on social media will never reach a patient's bedside.
How Long Does This Actually Take?
The average time from Phase 1 to regulatory approval is 10.5 years across all disease areas. But that number hides enormous variation.
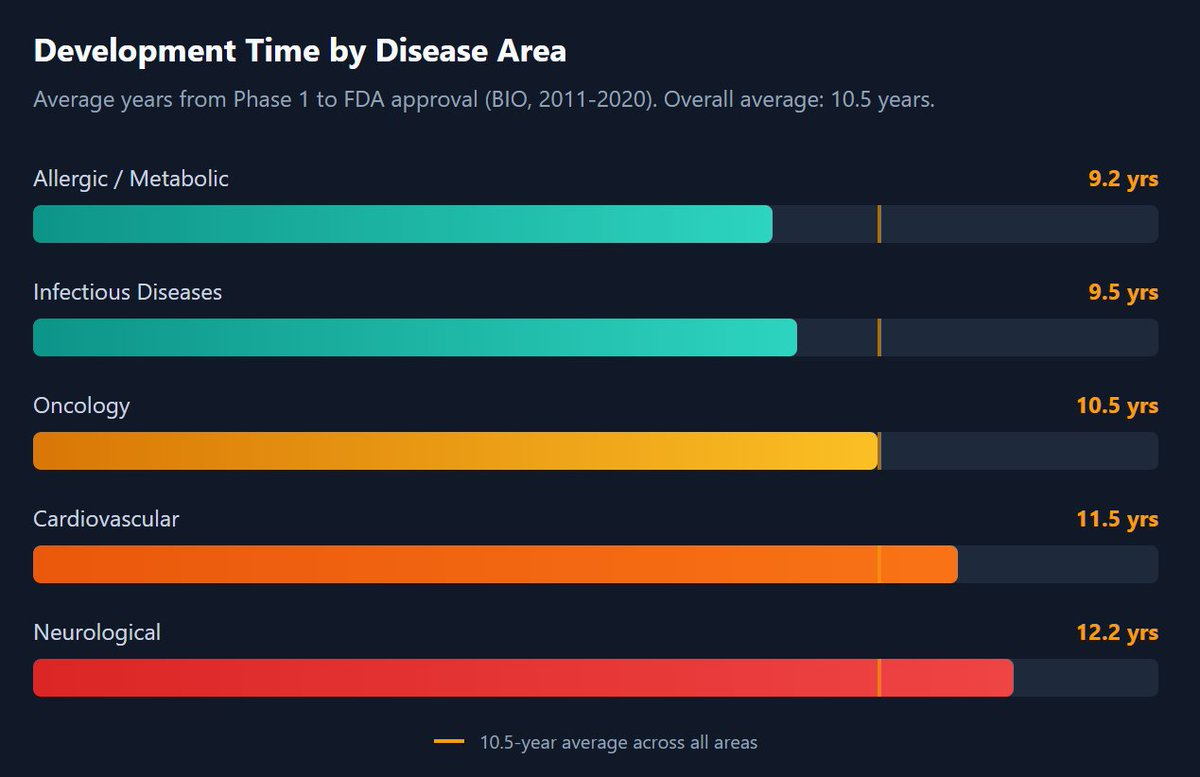
Neurological drugs take the longest because the brain remains one of the most complex and poorly understood organs. When someone tells you they've "cured" a neurological condition based on a Phase 1 result, they are, at best, 12 years and a 92% failure rate away from proving it.
The Bleeding Spray: A Case Study in PR Science
The specific "breakthrough" that sparked this article is a perfect example of how the PR Gap works.
The KAIST bleeding spray, technically called AGCL powder, uses alginate and chitosan to turn blood into a gel in one second. The technology is genuinely fascinating. But as of early 2026, here is its actual status:
It is in the pre-clinical and validation phase. It has been tested on animal models only. It has not received approval for widespread human clinical use or commercial sale.
Why This Story Went Viral: The Anatomy of PR Science

The researchers are highlighting its "potential" for civilian and emergency use specifically to attract the partnerships and funding needed to move into those grueling Phase 2 and 3 human trials. Without that PR noise, a project like this might sit in a lab for another decade.
There is a massive leap between "stopped a rabbit's liver from bleeding in a controlled lab" and "safe for a paramedic to spray into a human chest cavity in the rain."
What Real Progress Actually Looks Like
While the hype is loud, genuine advancement is happening in the shadows of "boring" Phase 3 successes. If you want to know what medicine is actually advancing in 2026, stop looking at viral tweets and start looking at the projects that have survived the validation process.
Proven Breakthroughs of 2025/2026: The Ones Nobody Tweeted About

These are the uhuru moments. Not because they look dramatic in a 15-second video, but because they have endured years of rigorous testing and emerged validated.
The Viral Spray vs. Proven Medicine: A Side-by-Side Comparison

It's easy to get excited about the spray because it looks like magic in a video. But the real "heroics" are happening in boring-sounding Phase 3 reports. That is where lives are actually saved.
The AI Paradox
AI is genuinely changing the game in drug discovery, and it would be dishonest to pretend otherwise. With AI-driven platforms, we are finding possible cures faster than ever. Computational models can simulate how a molecule interacts with a protein in milliseconds. The discovery phase that used to take years is being compressed into months.
But "possible" is the operative word.
A computer can predict that a molecule will stop bleeding. It cannot predict how a human immune system might react over six months. It cannot account for the infinite biological variability across age groups, ethnicities, pre-existing conditions, and genetic profiles.
Where AI Helps vs. Where It Hits a Wall
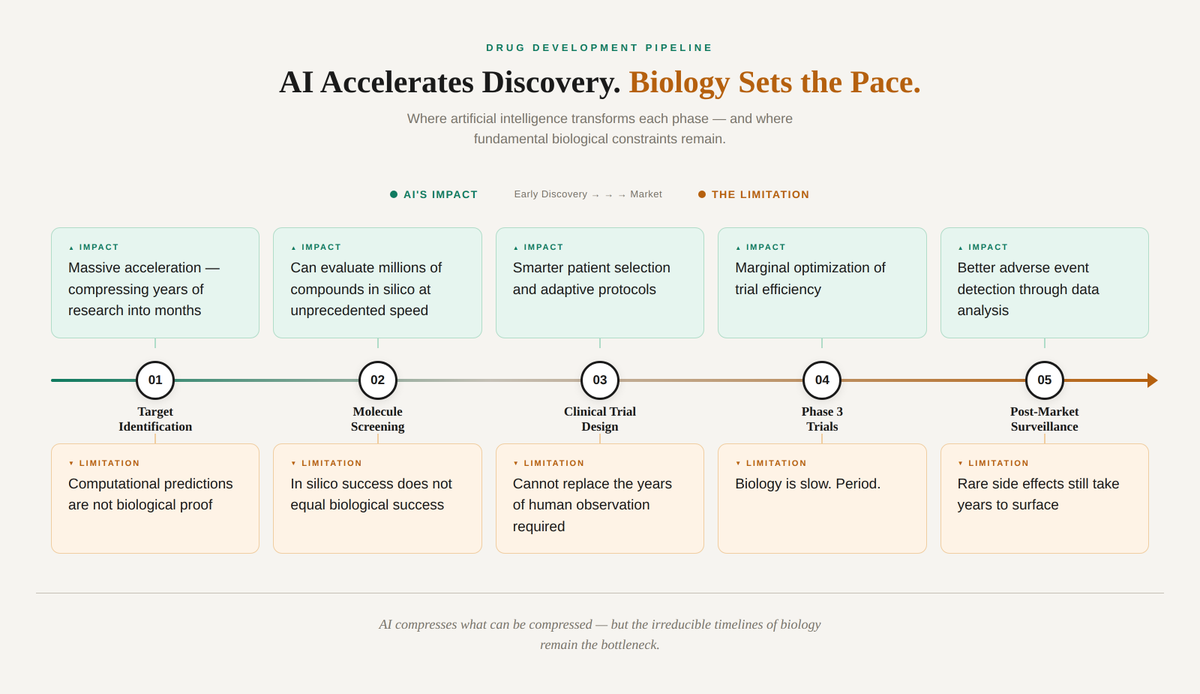
We are in a "hurry up and wait" era for medicine. The ideas come fast. The validation remains slow, human, and expensive. No amount of computational speed bypasses the biological reality that you have to test things in actual humans over time.
Even After Approval, The Wait Continues
Here is the part that almost nobody talks about. Let's say a drug beats every odd. It survives the 10.5-year average development timeline. It clears Phase 3. It gets FDA approval. The uhuru moment arrives.
Now what?
The uncomfortable truth is that even approved, validated treatments can take years or decades to become standard practice. Regulatory approval is not the finish line. It is the starting line of an entirely different race: the race to adoption.
The 17-Year Gap
In 2001, the Institute of Medicine published a landmark report called Crossing the Quality Chasm. Its finding sent shockwaves through the medical establishment: it takes an average of 17 years for new knowledge generated by research to be incorporated into routine clinical practice. And even then, application is "highly uneven."
This figure, originally from a study by Balas and Boren (2000), has been cited over 2,200 times in academic literature as of 2025. More recent research from Washington University confirms the pattern: evidence-based programs routinely take 15 to 17 years to reach routine practice, delaying health gains for an entire generation.
The NIH's own strategic plan estimates that moving a new drug or medical device from conception to market takes 14 years and costs $2 billion. And that is just to get it approved. Getting doctors to actually prescribe it? That is another journey entirely.
Why Approved Treatments Sit on the Shelf

Historical Proof: Breakthroughs That Took Decades
This is not a new problem. Throughout medical history, the gap between discovery and widespread adoption has been staggeringly long.

Consider the case of Ignaz Semmelweis. In 1847, he demonstrated that hand washing with chlorinated lime solution could reduce maternal mortality from 18.3% to under 2% in obstetric wards. The evidence was overwhelming. Maternal death rates plummeted wherever his protocol was adopted.
The medical establishment's response? They rejected him. Prominent physicians like Rudolf Virchow refused to believe that invisible particles on their hands could kill patients. Semmelweis was driven from his post, ridiculed, and eventually committed to a mental asylum, where he died in 1865 at the age of 47. It would take another two to three decades, until Pasteur's germ theory and Lister's antiseptic surgery gained traction in the 1870s and 1880s, for hand hygiene to become standard medical practice.
Semmelweis's story is the most dramatic example, but the pattern repeats: penicillin was discovered in 1928 but took 17 years and a world war to reach mass production. The link between H. pylori bacteria and stomach ulcers was proven in 1982, but it took until the late 1990s for the medical community to widely abandon the old "stress and spicy food" theory and adopt antibiotic treatment as standard care.
The Real Timeline: Discovery to Your Medicine Cabinet
When you combine all three phases of the medical journey, the total timeline becomes sobering:

This means that a "breakthrough" you see trending on social media today, even in the best-case scenario where everything goes right, might not reach your local hospital as standard practice until the 2040s or 2050s. For the viral bleeding spray currently in pre-clinical trials? If it follows the average path, you are looking at the mid-2030s for potential approval and the 2040s for widespread adoption.
That is not pessimism. That is the math.
Why the Hype Is Dangerous
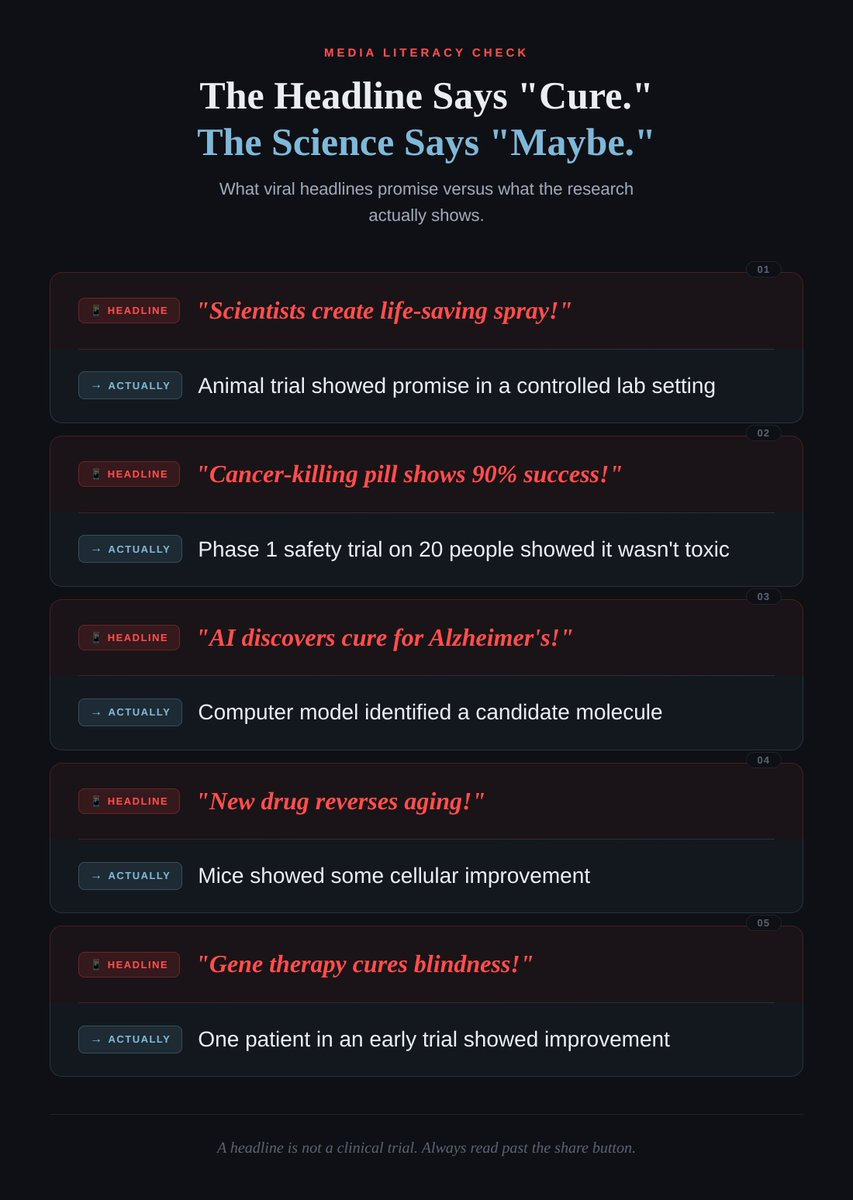
When a "one-second cure" goes viral, it creates a culture of science by press release. And the consequences are real.
False hope. Patients with chronic conditions see Phase 1 results and think a cure is months away, when it is usually five to ten years away. For someone managing a painful, debilitating illness, that kind of emotional whiplash is cruel.
Funding cannibalization. "Flashy" science, like a spray-on gel, often gets more attention and funding than slow, foundational research that nobody wants to tweet about but that underpins everything else.
Erosion of trust. When these viral breakthroughs inevitably fail to materialize, nobody runs the headline "Promising spray from 2024 didn't pan out in Phase 3." The silence erodes public confidence in science itself, making it harder to rally support when real breakthroughs do arrive.
The Hype Cycle vs. The Reality Cycle
The Geopolitical Pressure Cooker
This is not happening in a vacuum. The changing geopolitical landscape is squeezing healthcare from every angle. With interest rates fluctuating, government budgets reallocating toward defense spending, and traditional grant pools tightening, the financial ecosystem that once sustained patient, long-term research is fracturing.
Scientists are being forced to compete for attention in a media landscape that rewards hype over nuance. They shouldn't have to market their way to funding for legitimate research, but the alternative is watching their work die quietly in an underfunded lab.
The Funding Crisis by the Numbers

This is a systemic problem, not an individual failure. And until we fix the funding structures, the PR Gap will only widen.
Final Thought: Hold the Hope, Verify the Phase
It is okay to be excited about the one-second bleeding spray. It is brilliant engineering. But we must stop confusing discovery with delivery.
The next time you see a "miracle" on your timeline, ask yourself these three questions:
1. What phase is it in? If it's pre-clinical or Phase 1, you are looking at a minimum of 10 years before it could reach patients, and a greater than 90% chance it never will.
2. Has it been tested in humans? Animal models are necessary but insufficient. The leap from mouse to human is where most "miracles" fall apart.
3. Who is funding the announcement? If the researchers are using media coverage to attract investment, the story is as much about fundraising as it is about science.
Until a treatment clears Phase 3, it's not a breakthrough. It's a pitch.
And even after approval, the 17-year adoption gap means the road from "proven" to "available at your local hospital" is longer than most people realize. The full journey from a scientist's eureka moment to a treatment you can actually receive can span two to three decades.
While we hold space for the scientists doing incredible work under impossible conditions, we owe it to ourselves, and to the patients waiting for real cures, to demand honesty about where these discoveries actually stand.
The real heroes of 2026 medicine aren't in your viral feed. They're buried in Phase 3 reports that nobody shares, but everybody needs.
Sources and References:
- Biotechnology Innovation Organization (BIO), Clinical Development Success Rates 2011 to 2020
- Balas, E. A., & Boren, S. A. (2000), Managing Clinical Knowledge for Health Care Improvement
- Institute of Medicine (2001), Crossing the Quality Chasm
- Tufts Center for the Study of Drug Development (CSDD), 2018
- Nature, Clinical Development Times for Innovative Drugs (PMC, 2023)
- FDA CDER Annual Reports, 2008 to 2024
- National Institutes of Health Strategic Plan
- Northwestern Medicine, Research on Physician Adoption of New Treatments (2014)
- University Affairs, Closing the 17-Year Gap Between Scientific Evidence and Patient Care (2017)
- Washington University Implementation Science Communications (2026)
If this resonated with you, share it with someone who sent you a "miracle cure" link this week. The more we understand the process, the better we can support the science that actually saves lives.
Research-driven writing on healthcare, technology, policy, and the systems that shape care across Africa.













Member discussion